商品描述
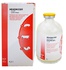
InoxelFor the treatment of diseases of bacterial etiology in cattle and pigs, prevention of bacterial infections in chickens < h3>General informationTrade name of the medicinal product: Inoxel.International nonproprietary name: ceftiofur.Dosage form: powder for the preparation of a solution for injection. Inoxel as an active substance in 1 bottle contains ceftiofur sodium-1.06 g (equivalent to 1 g of ceftiofur) or 4.2 g (equivalent to 4 g of ceftiofur).In appearance, the drug is an amorphous powder of white or light beige color.Inoxel is produced in the form of a sterile powder, packaged in 1.06 g or 4.2 g in glass vials with a capacity of 20 ml or 80 ml, respectively. The powder bottles are packed in individual cardboard boxes.Store Inoxel in the closed packaging of the manufacturer, separately from food and feed, in a dry place, protected from direct sunlight, at a temperature of 5 0C to 20 0C. The shelf life of the drug under the conditions of storage in a closed package is 2 years from the date of production, after opening the bottle – 28 days < br>It is forbidden to use Inoxel after the expiration date. Inoxel should be stored out of the reach of children.Unused medicinal product is disposed of in accordance with the requirements of the legislation. Pharmacological propertiesInoxel belongs to the antibacterial drugs of the cephalosporin group.Ceftiofur sodium, which is part of the drug, has a wide spectrum of antibacterial action on Gram-negative and gram-positive bacteria, including strains that produce beta-lactamase, and some anaerobic bacteria, including: Escherichia coli, Pasteurella (Mannheimia) haemolytica, Pasteurella multocida, Haemophilus somnus, Actinobacillus pleuropneumoniae, Haemophilus parasuis, Salmonella cholerasuis, Salmonells typhimurium, Streptococcus suis, Streptococcus zooepidemicus, Streptococcus equi, Streptococcus agalactiae, Streptococcus dysgalactiae, Streptococcus bovis, Pasteurella spp., Staphilococcus spp., Actynomyces pyogenes, Klebsiela, Citrobacter, Enterobacter, Bacillus spp., Proteus spp., Fusobacterium necrophorum and Porphiromonas assacharolytica (Bacteroides melaninogenicus). The mechanism of the bactericidal action of the antibiotic is to inhibit the synthesis of the cell wall of the bacterium. After parenteral administration, ceftiofur enters the systemic bloodstream, is rapidly metabolized, reversibly binds to plasma proteins, and is concentrated at the sites of inflammation.The maximum concentration of the antibiotic in the blood plasma after intramuscular administration of the drug is reached in 0.5-2 hours and remains at the therapeutic level for up to 24 hours. The antibiotic is excreted from the body mainly in the urine and partially with feces, the half-life, depending on the type, age of the animal and the method of administration, is 10-19 hours.< br> Inoxel by the degree of exposure to the animal body belongs to the moderately dangerous substances (hazard class 3 according to GOST 12.1.007). Application procedureInoxel is prescribed for the treatment of bacterial respiratory infections in cattle caused by Pasteurella haemolytica, Pasteurella multocida, Haemophillus somnus, Actinobacillus pleuropneumoniae, and pigs caused by Pasteurella haemolytica, Pasteurella multocida, Haemophillus somnus, Actinobacillus pleuropneumoniae, Salmonella cholerasuis, Streptococcus suis; for the treatment of necrobacteriosis associated with Fusobacterium necrophorum and Porphiromonas assacharolytica (Bacteroides melaninogenicus in cattle, and for the prevention of early death of one-day-old chickens caused by Escherichia coli and Staphylococcus aureus. Contraindication to the use of Inoxel is an increased individual sensitivity of the animal to antibiotics of the cephalosporin group.Inoksel used for animals parenterally in a sterile solution, making the vial 1.06 g sterile powder make 20 ml of solvent (water for injection, saline diluent for vaccines Marek (if the drug is used to treat birds)), in a bottle from 4.02 g of sterile powder 80 ml of solvent. Dissolution Inoksela should be carried out as quickly as possible, water for injection (water for injection, saline diluent for vaccines Marek (if the drug is used to treat birds)), type in a sterile syringe with a large amount of needle with a large diameter and contribute to the vial with powder, then shake the vial immediately until the powder is completely dissolved. The resulting therapeutic solution contains 50 mg of ceftiofur /ml. The term of use of the therapeutic solution is: when stored in a dark place at a temperature of 5°C to 20°C – 12 hours, when stored in the refrigerator at a temperature of 2°C to 8°C-7 days, when stored in frozen form at a temperature of minus 4 ° C to minus 18°C-8 weeks.The therapeutic solution is administered to the animal once a day in the following doses: - to cattle-intramuscularly 1 ml per 50 kg of weight (1 mg of ceftiofur per 1 kg of animal weight). The course of treatment for respiratory diseases caused by Mannheimia haemolytica, Pasteurella multocida and Histophilus somni – 3-5 days; for acute interdigital necrobacillosis (panaritium, hoof rot) caused by Fusobacterium necrophorum and Bacteroides melaninogenicus – 3 days; - pigs – intramuscularly 1 ml per 16 kg of weight (3 mg ceftiofura per 1 kg of animal weight). The course of treatment for respiratory diseases caused by Pasteurella multocida, Actinobacillus pleuropneumonia and Streptococcus suis is 3-5 days; - one-day-old chickens-subcutaneously in the neck at a dose of 0.2 ml. Before administration to chickens, the standard solution (50 mg of ceftiofur/ml) is diluted with water for injection in accordance with the table, so that 0.2 ml of the administered solution contains 0.08 – 0.2 mg of ceftiofur (3 column of the table).TableDose of ceftiofur/chicken Volume of standard Inoxel solution (50 mg / ml) Volume of added solvent1 2 30.08 mg 1 ml 124 ml < br>0.10 mg 1 ml 99 ml < br>0.12 mg 1 ml 82 ml0.14 mg 1 ml 70 ml0.16 mg 1 ml 61 ml0.18 mg 1 ml 54 ml0.20 mg 1 ml 49 mlWith an overdose of the drug, the animal may experience a decrease in appetite, inflammatory reaction at the injection site.Features of the action at the first administration of the drug and at its cancellation were not revealed.Skipping the next dose of the drug should be avoided, as this can lead to a decrease in therapeutic effectiveness. In case of missing one dose, it is necessary to introduce it as soon as possible, then the interval between injections of the drug does not change.Side effects and complications when using Inoxel in accordance with this instruction, as a rule, are not observed. In some animals, a local reaction may occur at the injection site in the form of edema, which resolves spontaneously within a few days. With increased individual sensitivity to the drug and the manifestation of allergic reactions, the use of the drug is discontinued and the animal is prescribed antihistamines and symptomatic therapy.Inoxel should not be prescribed simultaneously with drugs of bacteriostatic action, due to a decrease in its bactericidal activity, as well as mixed in one syringe with other drugs.Slaughter of cattle for meat is allowed no earlier than 24 hours, pigs-no earlier than 48 hours after the last application of Inoxel. The meat of animals that were forcibly killed before the expiration of these terms can be used as food for fur-bearing animals. Milk of dairy cows during and after the use of Inoxel is allowed to be used for food purposes without restrictions. Personal prevention measuresWhen working with Inoxel, you should follow the general rules of personal hygiene and safety, provided for when working with medicines. At the end of the work, wash your hands with warm water and soap.In case of accidental contact of the drug with the skin or mucous membranes of the eye, they should be washed with plenty of water. People with hypersensitivity to the components of the drug should avoid direct contact with Inoxel. In case of allergic reactions or accidental ingestion of the drug in the human body, you should immediately contact a medical institution (you should have the instructions for using the drug or the label). Empty vials from under the drug may not be used for household purposes, they are subject to disposal with household waste.Manufacturer:< / h3> < p>VETANCO S. A.; Chile 33 B 1603 CMA Vicinte Lopez, Buenos Aires, Argentina.
详情
同类产品
Gvardeyskie Korma LLC
Россия
Specify the price
USD
0
0
廠商批發銷售! 10袋起批發!!! 全價食品依 GOST R 55453-2022 生產。 保證指標及營養價值(g/100g):粗蛋白質量分數:19.2 精胺酸:1.41 組胺酸:0.58 異亮胺酸:0.73 亮胺酸:1.67 離胺酸:1.17 蛋胺酸:0.37 蛋胺酸+胱氨酸:0.82 苯丙胺酸:0.96 苯丙胺酸+酪胺酸:1.26蘇胺酸:0.79 色胺酸:0.25 纈胺酸:1.12 粗脂肪質量分數:13.4 亞麻油酸:1.53 鈣:1 .6磷:1.2 鈣磷比:4/3 鉀:0.7 鈉:0.15 氯化物:0.17 鎂:0.19 銅:3毫克鐵:10 毫克錳:11 毫克硒:53 微克鋅:14 微克維生素A:10,000 IU 維生素E :30 IU 維生素D3:1500 IU 新生產線,與領先獸醫合作開發的飼料配方。 該工廠位於地址:Gvardeysky區Kalinkovo村22B。 適合大中型犬種的全價食品,採用天然成分,採用當地原料製成,並添加了專為狗的飲食選擇的複合維生素。
GLOBAL WHOLESALE TRADING
Соединенные Штаты Америки
USD
140.0/个/件/只
140.0
GMP工廠供應高品質粉末皮膚曬黑肽10mg MT2 Melanotan II肽庫存Melanotan II CAS 121062-08-6 銷售MT2網上白色粉末高純度大庫存皮膚曬黑鼻噴霧MELANOTAN2批發價MT2鼻曬黑劑批發價melanotan2 10mg /瓶我們支援標誌設計;標籤服務產品名稱:Melanotan-II凍乾粉1)melanotan ii 別名:Melanotan,MT22)melanotan ii CAS No.:121062-08-63)melanotan ii 純度:99%.4)melanotan ii 分子式:C50H69N15O95)melanotan ii 分子量:1024.156)melanotan ii 包裝(注射劑):10mg 2cc 小瓶,10mg/瓶,10 瓶/盒7)melanotan ii 儲存:保護陽光陽光和2-8 攝氏度形式,Mela notan II (MT2) 小瓶應在冰箱溫度(2-8 攝氏度)下儲存,在該溫度下可保持穩定至少 12 個月。與溶液重新配製(混合)後,小瓶應儲存在冰箱中。 或者,您可以將所需劑量預先裝入註射器中並冷凍它們 - 然後當您想要注射劑量時,只需解凍一個注射器(需要幾分鐘)不要每次您想要注射時都冷凍/解凍 mt2 小瓶射擊,因為這會降低溶液的品質。在任何情況下都不應搖晃小瓶
БТ-2007
Алматы, Казахстан
USD
10.0/t
10.0
紅花粕作為育肥動物主要日糧的植物性蛋白質補充劑,可部分取代棉粕。 它的纖維含量為30-37%,是蛋白質的來源,蛋白質含量超過18%,還含有鈣、磷和鐵。
Veterinary drugs Simparika
Россия
USD
21.77 - 54.44/个/件/只
午安 我的名字是Daria,來自土耳其的商品🇹🇷「我們公司專門供應來自土耳其的商品。我們「直接」與製造商和供應商合作,這使我們能夠以最優惠的價格為客戶提供優質的商品。我們還處理包括物流、清關和品質控制。立即聯繫我們以了解更多資訊並開始合作。我們目前正在與獸藥合作,例如: - PREVICON 57、227 - Nexgard S、M、L、XL - Nexgard Spectra XS、 S ,N,L,XL。- Simparika 5(3 片),20(5-10 公斤),80(20-40 公斤)。我將根據要求發送價格表。交付 SDEK 貨到付款。我們的倉庫位於莫斯科,可以來倉庫購買。如果缺貨,土耳其發貨15-20天。
EUROPEAN FEEDING TECHNOLOGIES LLP
Казахстан
USD
0.5/t
0.5
“EUROPEAN FEEDING TECHNOLOGIES” LLP 公司是哈薩克市場最大的飼料和精礦製造商和供應商之一。 我們為所有類型的農場動物(包括牛、家禽等)創建了「SMARTFEED」品牌配合飼料。我們保證產品的高品質以及為畜牧業提供高成果的最佳飼料成分。 由於產品品質高且品種齊全,“EUROPEAN FEEDING TECHNOLOGIES” LLP 公司是許多農場和農業企業的可靠合作夥伴。 我們的飼料廠的推出考慮了所有全球要求和標準。 在我們的工作中,我們使用MK Technex LLC的設備,這保證了產品的可靠性和品質。我們的飼料包括來自法國公司Mg2Mix的預混料,該公司在預混料的品質和穩定性方面處於世界領先地位。
APM AZİM GIDA A.Ş.
Турция
USD
1.0 - 15.0/个/件/只
APM Shakarlama 是属于第二类的饼干:酥脆的沙质基底、硬坚果、粘稠的干浆果和精致芳香的巧克力甘纳许。
这些类别的供应商
此类别中的所有供应商
商品:
供应商:
最少订单:
经理处
销售部门:
销售部门:
销售部门:
信息
告诉经理,您在Qoovee 上找到了商品
供应商联系人不可用!
写信给供应商


